Understanding Electricity : Part 1
In this blog post I would like to explain the overall basics of what Electricity is and how it is made, as I currently understand it to be.
Energy
When most people think of electricity, they often think of it as being Energy. But what is energy?
Energy is defined as being the ability to do work. In order to perform any kind of work, energy must be involved or expended (converted from one form to another). Energy supplies the required force, or power, whenever any work is accomplished.
One form of energy is that which is contained by an object of motion. For example, whenever a hammer is set in motion in the direction of a nail, it possesses energy of motion. As the hammer strikes the nail (hopefully, and not your thumb), then the energy of motion is converted into work as the nail is driven into the wood. Even if you hit your thumb instead of the nail, the energy of motion of the hammer is converted into work as the hammer smashes your thumb (ouch). The distance the nail is driven into the wood depends on the velocity of the hammer at the time it strikes the nail.
Energy contained by an object due to its motion is called Kinetic Energy.
Now assume that the hammer is suspended by a string in a position several feet above the nail. As a result of gravitational attraction of the Earth on the hammer, the hammer will experience the forces of gravity pulling it downward. If the string is suddenly cut or broken, the force of gravity will pull the hammer downward against the nail, driving it into the wood. While the hammer is suspended over the nail it has ability to do work because of its elevated position in the Earth's gravitational field. Since energy is the ablity to do work, the hammer contains energy.
Energy contained by an object due to its position is called Potential Energy.
The amount of potential energy available is equal to the product of the force required to elevate the hammer and the height that it is elevated.
Another example of potential energy is the energy contained within a tightly coiled spring. The amount of energy released when the spring unwinds depends on the amount of force required to wind the spring initially.
Electrical Charges
By studying the properties of electrostatics we see that a field of force exists in the space surrounding any electrical charge. The strength of the field is directly depended on the force of the charge.
The charge of one electron might be used as a unit of electrical charge, since charges are created by displacement of electrons. But keep in mind that the charge of one single electron is so small that it is impractical to use by itself for any real world applications.
The practical unit adopted for measuring charges is the Coulomb. One Coulomb is equal to the charge of 6,280,000,000,000,000,000 (six quintillion two hundred eighty quadrillion) (or 6.28 x 10^18) electrons (or to state it in another way, that is a quantity of electrons that is equal to 6.28 times 10 to the 18th power).
When a charge of one coulomb exists between two bodies, one unit of electrical potential energy exists, which is called the difference of potential between the two bodies. This is referred to as Electromotive Force or more commonly known as Voltage, and the unit of measure is the Volt.
Electrical charges are created by the displacement of electrons, so that there exists an excess of electrons at one point, and a deficiency at another point. Consequently, a charge must always have either a negative or positive polarity. A body with an excess of electrons is considered to be negative, because the quantity of electrons (which are negative in charge) would outnumber the quantity of protons (which are positive in charge). Whereas a body with a deficiency of electrons is positive, because the quantity of protons (which are positive in charge) outnumber the quantity of electrons (which are negative in charge).
A difference of potential can exist between two points, or bodies, only if they have different charges. In other words, there is no difference in potential between two bodies if both bodies have a deficiency of electrons to the same degree. If, however, one body is deficient of 6 coulombs (representing 6 volts), and the other body is deficient by 12 coulombs (representing 12 volts), then there is a difference of potential of 6 volts between the two bodies. The body with the greater deficiency (the least amount of electrons) is positive with respect to the other body.
In most electrical circuits only the difference of potential between two points is of importance and the absolute potentials of the points are of little concern. Very often it is convenient to use one standard reference for all of the various potentials throughout a piece of equipment. For this reason, the potentials at various points in a circuit are generally measured with respect to the metal chassis on which all parts of the circuit are mounted. The chassis is considered to be at zero potential and all other potentials are either positive or negative with respect to the chassis. When used as the reference point, the chassis is said to be at Ground Potential. For most common residential appliances and electronic systems, the Earth itself is often used as the Ground Potential.
Occasionally, rather large values of voltage may be encountered, in which case the volt becomes too small of a unit for convenience. In a situation of this nature, the kilovolt (kV) is used, meaning 1,000 volts. In other cases, the volt may be too large a unit, as when dealing with very small voltages. For this purpose the millivolt (mV) meaning one-thousandth of a volt, and the microvolt (µV) meaning one-millionth of a volt, are used instead. For example 0.001 volt would be written as 1 mV, and 0.000025 volt would be written as 25 µV.
When a difference in potential exists between two charged bodies that are connected by a conductor, electrons will flow along the conductor. This flow is from the negatively charged body (which contains an excess of electrons) to the positively charged body (which is deficient in electrons as compared to the negatively charged body). The flow between them will continue until the two charges are equalized and the potential difference no longer exists. An analogy of this is to visualize two water tanks connected by a pipe and valve. At first the valve is closed and all of the water is in tank A. Thus, the water pressure across valve is at maximum. When the valve is opened, the water flows through the pipe from tank A to tank B until the water level becomes the same in both tanks. The water then stops flowing in the pipes, because there is no longer a difference in water pressure between the two tanks.
Electron movement through an electric circuit is directly proportional to the difference in potential or electromotive force (emf), across the circuit, just as the flow of water through the pipe is directly proportional to the difference in water level in the two tanks.
A fundamental law of electricity is that the Electron Flow Is Directly Proportional To The Applied Voltage. If the voltage is increased, the flow is increased. If the voltage is decreased, the flow is decreased.
How Voltage Is Produced
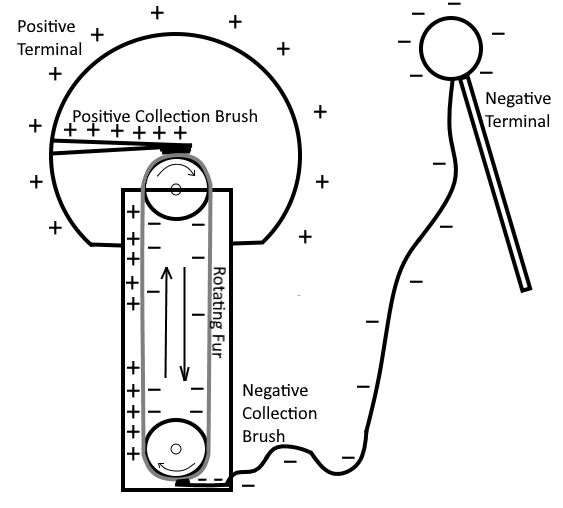
Again by studying the properties of electrostatics we can demonstrate that a charge can be produced by rubbing a rubber rod with fur. Because of the friction involved, the rod acquires electrons taken from the fur, resulting in an excess of electrons to build up on the rod, making it negative. The fur itself becomes positive (relative to the charge on rod) due to the loss of electrons. These quantities of charge constitute a difference of potential to know exist between the rod and the fur. The electrons which make up this difference of potential are capable of doing work if a discharge is allowed to occur (if the electrons in the body that has an excess of electrons are allowed to flow to the body that has less electrons).
To be a practical source of voltage, the potential difference must not be allowed to dissipate, but must be maintained continuously. As one electron leaves the concentration of negative charge, another must be immediately provided to take its place or the charge will eventually diminish to the point where no further work can be accomplished.
a Voltage Source is a device which is capable of supplying and maintaining voltage while some type of electrical apparatus is connected to its terminals. The internal action of the source is such that electrons are continuously removed from one terminal, keeping it positive, and simultaneously supplied to the second terminal which maintains a negative charge.
Presently, there are six known methods for producing a voltage or electromotive force (emf). Some of these methods are more widely used than others, and some are used mostly for specific applications:
- Friction - voltage produced by rubbing certain materials together. Static Electricity is one example of producing voltage by the act of friction.
- Pressure - (piezoelectricity) voltage produced by squeezing crystals of certain substances.
- Heat - (thermoelectricity) voltage produced by heating the joint (junction) where two unlike (dissimilar) metals are joined.
- Light - (photoelectricity) voltage produced by light striking photosensitive (light sensitive) substances.
- Chemical Action - voltage produced by chemical reactions, such as in a battery cell.
- Magnetism - Voltage produced in a conductor when the conductor moves through a magnetic field, or a magnetic field moves through the conductor in such a manner as to cut the magnetic lines of foce of the field.
Friction
It is believed that friction is the first method discovered for creating a voltage, in the very early days of human history. The development of charges produced by rubbing a rod with fur is a prime example of the way in which a voltage is generated by friction. However, because of the crude nature of the materials with which this voltage is generated, it cannot be conveniently used or maintained for practical prolonged use. For this reason, very little practical use has yet to be found for voltages generated by this method, since the method itself is difficult to maintain a continuous stable output.
In the search for methods to produce a voltage of a larger amplitude and of a more practical nature that still relies on this principle of friction, machines were developed in which charges were transferred from one terminal to another by means of rotating glass discs or moving belts and allowing them to rub onto hair or fur. The more notable of these machines is the Van de Graaff generator. It is used today to produce potentials in the order of Millions of Volts, and is commonly used for nuclear research and lab experiments.
| Van de Graaff Generator |
Continue with Electricity - Part 2