Understanding Electricity : Part 2
Continued from Electricity - Part 1...
Pressure
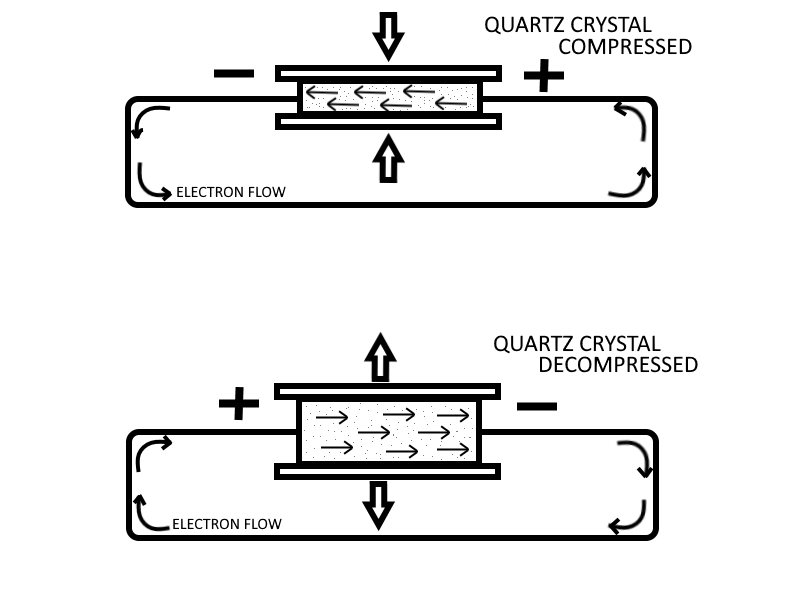
One specialized method of generating an emf utilizes the characteristics of certain ionic crystals such as quartz, Rochelle salts, and tourmaline. These crystals have the remarkable ability to generate a voltage whenever stresses are applied to their surfaces. Thus, if a crystal of quartz is squeezed, charges of opposite polarity will appear on two opposite surfaces of the crystal. If the force is reversed and the crystal is stretched, charges will again appear, but will be of the opposite polarity from those produced by squeezing. If a crystal of this type is given a vibratory motion, it will produce a voltage of reversing polarity between two of its sides. Quartz or similar crystals can thus be used to convert mechanical energy into electrical energy. This phenomenon is called the Piezoelectric Effect.
Some of the common devices that make use of piezoelectric crystals are microphones, phonograph cartridges, and oscillators used in radio transmitters, radio receivers, and sonar equipment. This method of generating an emf is not suitable for applications having large voltage or power requirements, but is widely used in sound and communications systems where small signal voltages can be effectively used.
| Piezoelectric |
It should be noted that the voltage produce is only produced while a change in the pressure (or vacuum) difference is applied and exerted (while the difference amount is changing, not while at rest). Once an amount of pressure (or vacuum) is established and maintained then the crystal will stop producing voltage, and it will only start producing voltage again once a change in difference of pressure (or vacuum) occurs.
Crystals of this type also possess another interesting property, the "converse piezoelectric effect". That is, they have the ability to convert electrical energy into mechanical energy. A voltage impressed (applied) across the proper surfaces of the crystal will cause it to expand or contract its surfaces in response to the voltage applied. This is commonly used in sound buzzers, and small electric horns.
Heat
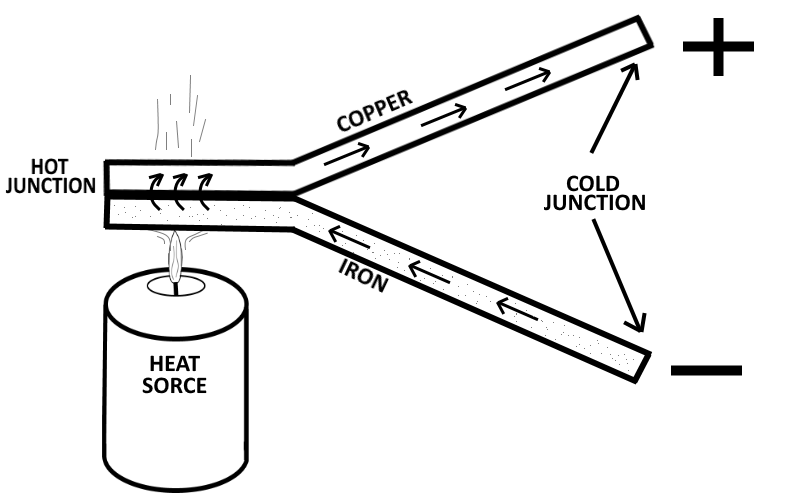
When a length of metal (no matter if it is in the form of wire, plate, or sheets), is heated at one end, electrons tend to move away from the hot end toward the cooler end. This is true of most metals. However, in some metals, such as iron, the opposite takes place and electrons tend to move toward the hot end instead of move away from the hot end.
| Thermocouple |
If you connect one end of a copper wire to one end of an iron wire, and then heat the the conjunction (the point of connection of the two wires), then the electrons (the negative charges) will move through the copper away from the heat, and at the same time the electrons will move through the iron toward the heat. This produces an excess of electrons to build up on the open end of the copper wire (the cool end of the copper wire) and a lack of electrons to exist on the open end of the iron wire (the cool end of the iron wire). If a current meter (known as an Ammeter) is connected between the cool junctions (the open ends of the wire), then an electron flow is detected moving from the copper wire to the iron wire through the current meter. This device (two dissimilar metals connected together on one end) is generally referred to as a Thermocouple. Thermocouples have somewhat greater power capacities than crystals, but their capacity is still very small if compared to some other sources.
The thermoelectric voltage in a thermocouple depends mainly on the difference in temperature between the hot and cold junctions. The greater the difference in temperatures, the greater the voltage output will be.
Thermocouples are widely used in automatic temperature control equipment as sensors because they can convert temperature heat into electricity, and they can generally be subjected to much greater temperatures than ordinary thermometers (such as the mercury or alcohol types).
Another use of thermocouples on a much larger scale is in the production of electricity in harsh environments such as deep space satellite systems and Mars rovers, by converting the temperature differences of internal heat generated from nuclear sources with external coldness of outer space.
Similar to the converse piezoelectric effect, by applying voltages to certain types of thermocouples, hot and cold effects can be produced by as much as 40 F degrees differences (one side will get hot while the other side gets cold, and the difference between them can be about 40 F degrees). These TEC thermocouple system are often used with computer CPU's to help keep the CPU's from overheating, as well as solid state refrigerator systems.
Light
When a photon of light strikes the surface of a substance, it may dislodge electrons from their orbits around the surface atoms of the substance. This occurs because light has energy, the same as any moving force.
Some substances, mostly metallic ones, are far more sensitive to light than others. That is, more electrons will be dislodged and emitted from the surface of a highly sensitive metal, with a given amount of light, than will be emitted from a less sensitive substance. Upon losing electrons, the photosensitive (light-sensitive) metal becomes positively charged, and an electric force is created. Voltage produced in this manner is referred to as a Photoelectric Voltage.
The photosensitive materials most commonly used to produce a photoelectric voltage are various compounds of silver oxide or copper oxide. A complete device which operates on the photoelectric principle is referred to as a photoelectric cell. There are many different sizes and types of photoelectric cells in use, and each serves the special purpose for which it is designed, but they all still have very similar basic features.
When light strikes the sensitive surface, it emits electrons toward an anode (a conductor that collects the emitted electrons). The more intense the light, the greater the number of electrons emitted. When a wire is connected between the anode and the dark side of the cell (the back side), the accumulated electrons will flow to the dark side. These electrons will eventually pass through the metal of the reflector and replace the electrons leaving the light sensitive surface. Thus, light energy is converted to a flow of electrons, and a usable current is developed.
For example, if you coat a base plate of pure copper with light sensitive copper oxide, and place an extremely thin semitransparent layer of metal over the copper oxide, then the semitransparent layer will allow light to travel through it to the sensitive copper oxide and at the same time it will collect the expelled electrons from the sensitive copper oxide as the light collides with it. An external wire connected to the semitransparent surface to the back side of the copper oxide place completes the electron path. The photocell's voltage is used as needed by connecting the external wires to some other device, which amplifies (enlarges) it to a usable level.
The power capacity of a single photocell is very small. However, it reacts to light-intensity variations in an extremely short time. This characteristic makes the photocell very useful in detecting or accurately controlling a great number of operations. For instance, the photoelectric cell, or some form of its principle, is used in television cameras, cell phone cameras, web cameras, automatic manufacturing process controls, door openers, burglar alarms, and so forth.
Individual photocells can be connected together into an array, in efforts to make solar panels. By connecting several solar panels together a realistic quantity of voltage can be produce from sunlight that can be used to supply charging power to deep cell batteries, which can then be used to supply stable power to electronic devices.
Chemical Action
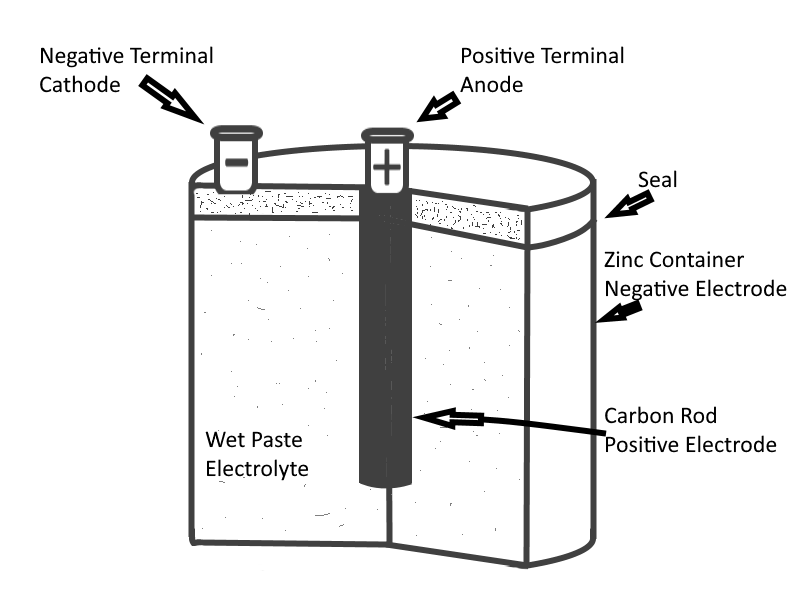
If two dissimilar substances (usually metals or metallic materials) are immersed in a solution that produces a greater chemical action on one substance than on the other, a difference of potential will exist between the two. If a conductor is then connected between them, electrons will flow through the conductor to equalize the charge. This arrangement is called a Primary Cell. The two metallic pieces are called Electrodes, and the solution is called the Electrolyte.
The difference of potential results from the fact that material from one or both of the electrodes goes into solution in the electrolyte. In the process, ions form in the vicinity of the electrodes within the solution as the solution chemically interacts with the electrodes substances. Due to the electric field associated with the charged ions, the dissimilar electrodes acquire dissimilar charges. In effect, the chemical reaction of the solution will strip away electrons from one electrode (causing that electrode to become Positive in charge), and accumulate those electrons onto the other electrode (causing that electrode to become Negative in charge).
The amount of difference in potential between the electrodes depends principally on the metals used. The type of electrolyte and the size of the cell actually have very little or no effect on the potential difference produced.
There are two types of primary cells:
- The dry cell : More commonly used, is not actually dry, but contains an electrolyte mixed with other materials to form a paste. Cellphone batteries and flashlight batteries are commonly powered by dry cells.
- The wet cell : A cell with a liquid electrolyte must remain in an upright position and is not readily transportable. An automotive battery is an example of this type of cell.
| Dry Cell |
Batteries are formed when several cells are connected together to increase electrical output.
Magnetism
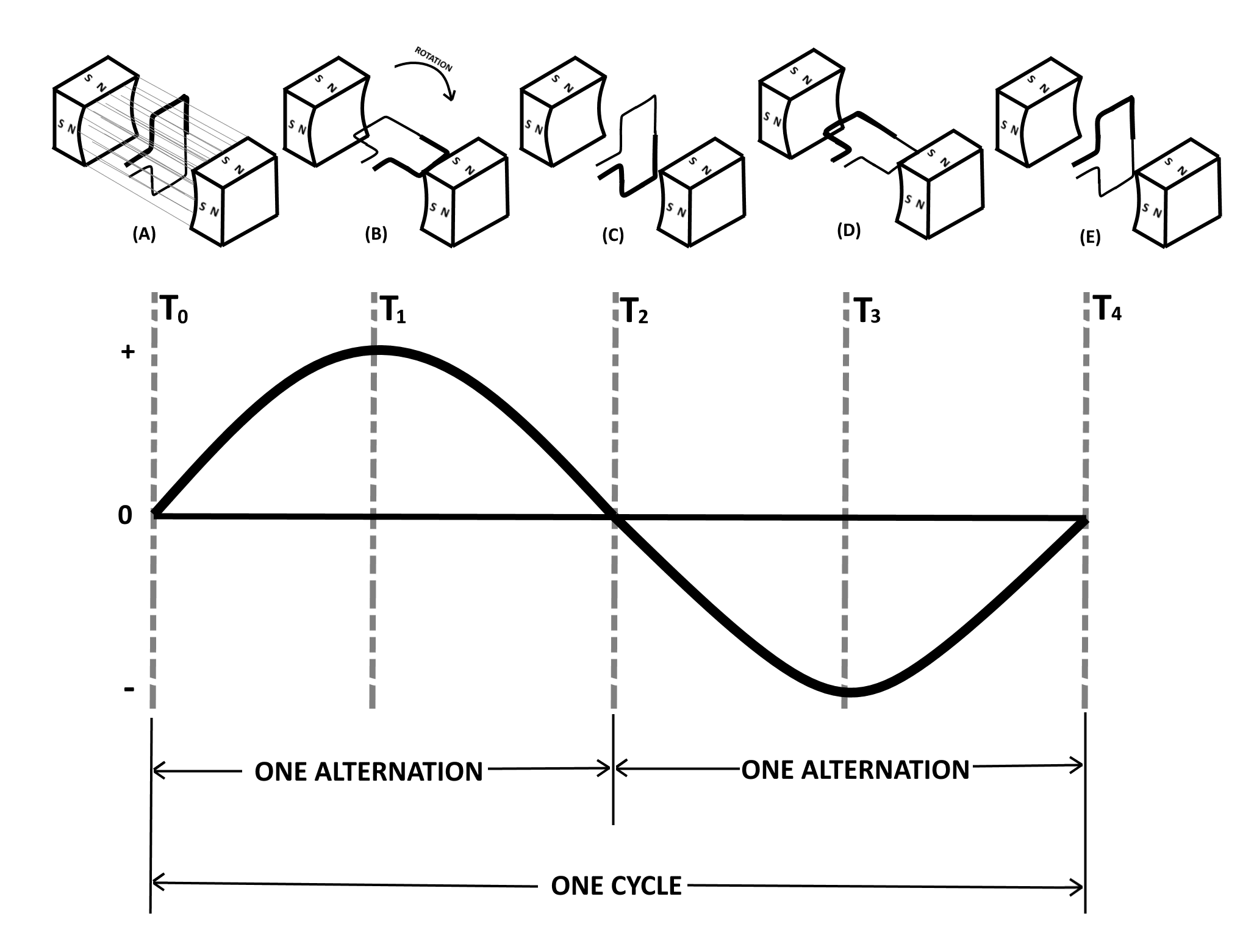
One of the most useful and widely employed applications of magnets is in the production of vast quantities of electric power from mechanical sources. The mechanical power may be provided by a number of different sources, such as gasoline or diesel engines, as well as from water, steam, or wind turbines. However, the final conversion of these source energies into electricity is done by generators employing the principle of electromagnetic induction.
The important note to take from this is that ALL such electromagnetic-induction generators follow the same overall basic principles:
- There must be a Conductor in which the voltage will be produced.
- There must be a Magnetic Field in the conductor's vicinity.
- There must be a relative Motion between the field and conductor. The conductor must be moved so as to cut across the magnetic lines of force, or the field must be moved so that the lines of force are cut by the conductor.
When a conductor or conductors move across a magnetic field so as to cut the lines of force of that magnetic field, electrons within the conductor are propelled in one direction or another, resulting in the electrons to be accumulated more on one end of the conductor and less on the other end. Thus, an electric force, or voltage, is created (generated) as a result of this electron difference within that conductor. The movement of the electrons occurs because of the magnetically induced emf acting on the electrons in the conductor.
| AC Generator |
If a path of electron flow is provided between the ends of the conductor, electrons will leave the negative end and flow to the positive end. Electron flow will continue as long as the emf exists. The emf can be created by moving the conductor so that it cuts through the lines of force of the magnet, as well as by holding the conductor still and moving the magnet so that its lines of force cut through the conductor and moving the magnetic field back and forth.
It should be noted that once the motion stops and the conductor no longer passes through the magnetic lines of force of the magnets, then the electron flow stops and it will no longer generate or produce electricity. Therefore to produce electricity, the conductor must remain in constant motion repeatedly cutting through the magnetic lines of force of the magnet.
Continue with Electricity - Part 3